
Hippo keygen

Hippo keygen
The latest versions
of the best software
Latest Software News
Microsoft announces when it will kill Wunderlist

Microsoft's Wunderlist to-do app has been living on borrowed time for a while now. It was back in 2017 when the software giant announced it would be phasing out the app, which strangely enough, Microsoft had only purchased in 2015.
Patrick Devaney - 9 months ago
Google launches Android Ambient Mode

Android's latest Google Assistant powered feature, Ambient Mode, will begin rolling out next week. Google first announced Ambient Mode back in September, telling the world that the feature would turn Android devices into smart devices whenever they're plugged in and charging.
Patrick Devaney - 10 months ago
Instagram unveils Reels, a TikTok clone
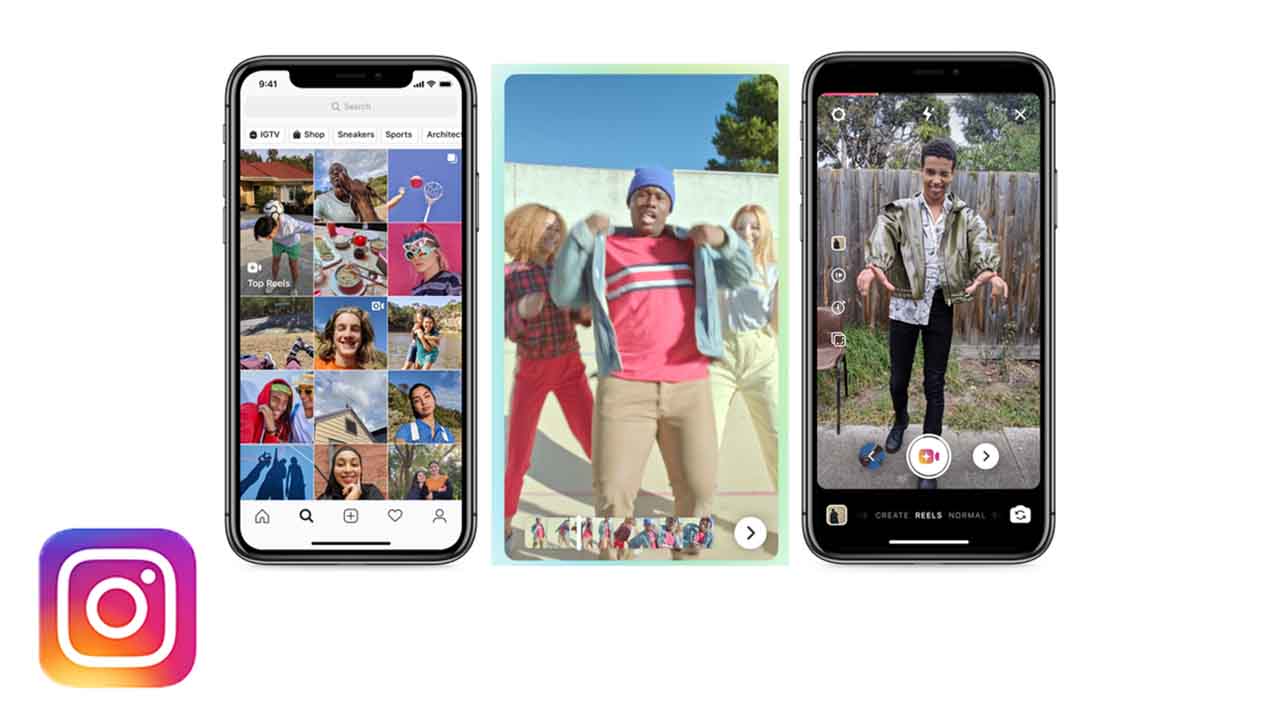
Is TikTok the best app in the world right now? There is an increasing amount of evidence that a lot of people at Facebook and Instagram HQ seem to think so.
Patrick Devaney - 10 months ago
What is Windows 10X?

Foldable screens and new dual screen devices have increasingly become the trend. Manufacturers now want to show themselves as forward thinking hardware developers. Microsoft's upcoming Windows 10X operating system has long been rumored to be the software giant's first foray into the dual screen world we all look likely...
Patrick Devaney - a year ago
Report: Your phone number reveals more personal info than your name

According to a recent New York Times article, your phone number has loads of personal information attached to it. Quite often, websites and apps ask us for our phone number when logging in or creating an account.
Jacob Yothment - a year ago
A dark mode is coming to Google Chrome on desktop

Every app these days seems to have a new dark mode or one in development. This is due in part to the appearance a slick black background can give but also due to recent research from Google that shows darker colors use up less battery power on devices with...
Patrick Devaney - a year ago
Google now allows you to play podcasts within search results

The podcasting world has always been a bit of the Wild West. You'd discover a series through word of mouth or maybe by way of an article listing recommendations. Then it's on you to use a podcasting app or go to the website to download the episode and listen..
Ben Bowman - a year ago
LEAK: Windows 10 removes live tiles in new Start menu
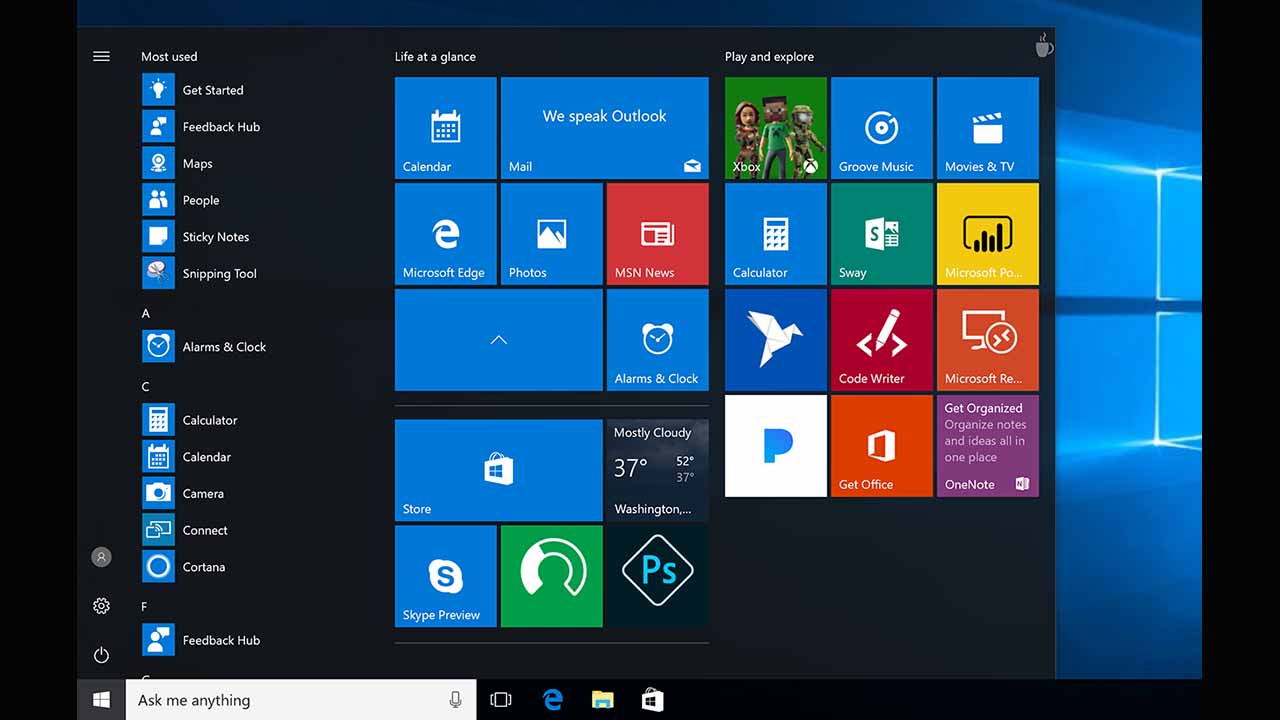
A new leak shows that Windows 10's new look will not feature live tiles. For those who do not know, live tiles are the moving tiles on your Start menu.
Jacob Yothment - a year ago
partition magic keygen At A Discount!
Amazing Magic Tricks and Magic Courses - Free Magic eBook - Learn How to do Magic Tricks of all kinds. Discover The Insider Secrets Of MAGIC That Will Amaze and Amuse You and Gain The Admiration and Respect of All Your Friends! - Guaranteed 100%! YOU'LL LOVE THE LOOK OF AMAZEMENT ON THEIR FACES and the ENTHUSIASTIC APPLAUSE YOU'LL GET.... EVERY TIME!" Discover the Magic Tricks and Secrets of professionals in the privacy and comfort of your own home... You'll get the Insider Secrets of How To Do.... close-up magic, card magic, coin magic, rope magic, street magic & cabaret magic! The best source of practical Professional Magic on the Internet - Where You'll Learn Real Magic and gain the admiration and respect of all your friends - - Everyone Loves Magic! REAL MAGIC TRICKS - THAT YOU CAN DO! Start Your Magic Journey Here for FREE....� Go To� partition magic keygen For More Details.
Find partition magic keygen at Amazon.com and get Free Shipping today!
To Find Other partition magic keygen 's
PartitionMagic Pro v7.0 French by FFF
CrackSpider.net. PartitionMagic Pro v7.0 French by FFF. Download the crack/serial/keygen for "PartitionMagic Pro v7.0 French by FFF". NFO ... PartitionMagic PRO 7.0 Fr ...
http://partition.crack.cd/Partition_Magic_Pro_v7.0_French_by_FFF.html
download free full magicpartition powerquest
We have download free full magicpartition powerquest tricks. Click here if you're looking for download free full magicpartition powerquest. ... crack acronis partitionmagic 2003. metapoisk.ru ... XP v7.21.1265 - download KEYGENPartitionMagic v8.0 ... TweakNow ... v7.01 PowerQuestPartitionMagic 8.0 Full ...
http://www.free-magic.com/magic/download-free-full-magic-partition-powerquest.html
gulli �|� Alle Ergebnisse zu partitionmagic �|� gulli
gulli �|�zu partitionmagic wird diese Undergroundseite empfohlen �|� underground-suche! ... PartitionMagic 7.0 - 1 ... Magic Phone v1.0 crack by DBC... ... v1.0 serial Partioner Partionstar v1.32 by ECLiPSE Partition Commander... ... v3.51 serial Partition Commander v8.02 keygen ...
http://search.gulli.com/partition+magic
search [stat]
...
CRB3 regulates contact inhibition by activating the Hippo pathway in mammary epithelial cells
Abstract
The loss of contact inhibition is a hallmark of cancer cells. The Hippo pathway has recently been shown to be an important regulator of contact inhibition, and the cell apical polarity determinant protein CRB3 has been suggested to be involved in Hippo signalling. However, whether CRB3 regulates contact inhibition in mammary cells remains unclear, and the underlying mechanisms have not been elucidated. As shown in the present study, CRB3 decreases cell proliferation, promotes apoptosis, and enhances the formation of tight and adherens junctions. Furthermore, we report for the first time that CRB3 acts as an upstream regulator of the Hippo pathway to regulate contact inhibition by recruiting other Hippo molecules, such as Kibra and/or FRMD6, in mammary epithelial cells. In addition, CRB3 inhibits tumour growth in vivo. Collectively, the present study increases our understanding of the Hippo pathway and provides an important theoretical basis for exploring new avenues for breast cancer treatment.
Main
Contact inhibition is a process by which cells stop proliferating when they contact adjacent cells. However, cancer cells do not arrest their growth when they interact with neighbouring cells. Instead, they continue to proliferate, grow on top of each other and form multilayered foci.1, 2, 3 The loss of contact inhibition is a hallmark feature of cancer cells.4 Therefore, contact inhibition is a powerful anticancer mechanism that prevents cell proliferation. Although a number of signalling pathways have been shown to be involved in contact inhibition, the underlying mechanisms remain poorly understood.
The Hippo pathway plays a crucial role in regulating contact inhibition and is highly conserved in species ranging from Drosophila to mammals as almost all the components of the pathway in Drosophila have recognizable homologues in mammals.5, 6, 7 Mst1/2 is activated during the activation of the Hippo pathway in mammals, allowing it to subsequently activate Sav1 and Mob1. Then, Lats1/2 kinases are phosphorylated and activated. Upon the activation of Lats1/2 kinases, the Lats1/2-Mob1 complex in turn phosphorylates YAP/TAZ. Lats1/2 regulate the transcriptional co-activators YAP/TAZ by altering their localization and protein stability. Phosphorylated YAP/TAZ are retained in the cytoplasm when bound to 14-3-3 and are degraded by β-TrCP.8, 9, 10 Upon inactivation of the Hippo pathway, the dephosphorylated YAP/TAZ translocate to the nucleus, where they bind to transcription factors such as TEAD and SMAD.11, 12 In addition to these downstream regulators, multiple upstream regulators of the Hippo pathway have been identified in Drosophila, including WW and C2 domain containing 1 (WWC1/Kibra, homologue of mammalian Kibra),13, 14, 15 Merlin (homologue of mammalian NF2) and Expanded (homologue of mammalian FRMD6).16, 17, 18 However, the upstream regulators have not been studied as intensively as the kinase cascade in mammals.
Apical-basal polarity components, particularly the apical polarity determinant Crb, have recently been suggested to regulate Hippo pathway activity in Drosophila.19 Three apical-basal polarity complexes have been identified to date, including the CRB (Crumbs) complex (CRB/PATJ/PALS1), PAR complex (PAR3/PAR6/aPKC) and Scribble complex (SCRIB/DLG/LGL). These complexes work together to maintain apical-basal polarity.20 Mammals have three Crumbs family members (CRB1-3).21 In Drosophila, Crb directly interacts with the upstream Hippo pathway component Expanded.22, 23, 24 In mammals, the depletion of CRB3 leads to increased nuclear localization of YAP/TAZ,25, 26 suggesting that CRB3 is associated with the Hippo pathway. Little is currently known about the functional roles of CRB3 in mammals. Furthermore, CRB3 is associated with contact inhibition.21 Nevertheless, the detailed molecular mechanisms linking CRB3 to the regulation of the Hippo pathway and contact inhibition are still not clear.
In this study, we aimed to investigate the precise mechanisms by which CRB3 regulates the Hippo pathway and contact inhibition. Based on our observations, CRB3 acted as an upstream regulator of the Hippo pathway and regulated contact inhibition via Kibra and/or FRMD6.
Results
CRB3 is an important mediator of contact inhibition
We examined CRB3 expression in breast cancer tissues and cells to investigate the role of CRB3 in breast cancer. CRB3 expression was low in breast cancer tissues but was high in adjacent normal breast tissues (Supplementary Figures S1a–c). CRB3 expression was correlated with tumour size but not with age, histological grade, clinical stage or lymph node involvement (Supplementary Table S1). The expression levels of the CRB3 mRNA and protein were significantly lower in breast cancer cells than in immortalized mammary epithelial cells (Supplementary Figures S1d and e). Our results may provide a clue about the relationship between the loss of CRB3 expression and the loss of contact inhibition, a hallmark of cancer cells.4
CRB3 expression was examined in cells grown under sparse and confluent conditions to determine the relationship between CRB3 expression and contact inhibition. CRB3 expression was significantly lower in breast cancer cells than in immortalized mammary epithelial cells, regardless of cell density (Figures 1a and b). Interestingly, CRB3 expression was significantly lower in confluent MCF10A and MCF12A cells than in the sparse cells, but further studies are needed to investigate the mechanisms underlying the contact-dependent change in CRB3 expression. The localization of CRB3 is essential for the maintenance of cell polarity. Immunofluorescence staining for CRB3 showed that the protein was predominantly expressed in the membrane and cytoplasm of sparse MCF10A cells and sparse and confluent MDA-MB-231 cells, but CRB3 was predominantly localized in a certain region of the membrane of confluent MCF10A cells (Figures 1c and d). The different localization patterns of CRB3 in confluent cell cultures may suggest that its function varies among different cells.
CRB3 was knocked down in MCF10A cells and overexpressed in T47D and MDA-MB-453 cells (Figures 1e and f and Supplementary Figures S2a–b). Cell proliferation assays, cell cycle analyses and BrdU incorporation assays were performed to examine the effects of CRB3 on cell proliferation. CRB3 knockdown accelerated G1/S progression and increased BrdU incorporation and cell proliferation in MCF10A cells grown under sparse conditions, whereas CRB3 overexpression decreased BrdU incorporation and cell proliferation in T47D and MDA-MB-453 cells, regardless of cell density (Figures 1g–j, Supplementary Figures S3a–b and Supplementary Figures S2c–e). However, CRB3 overexpression in T47D cells had no effect on the cell cycle, except that it induced the appearance of a pre-G1 peak (Figure 1h and Supplementary Figures S3c). Thus, CRB3 plays an important role in contact inhibition. Moreover, the expression levels of p27 and p53, two regulators of contact inhibition,27 were decreased in shCRB3 cells (Figure 1k). In addition, p16, another regulator of contact inhibition,27, 28 was not expressed because MCF10A cells may have p16 deletion, as previously reported by Debnath J et al.29 The levels of p27, p53 and p16 were increased when CRB3 was overexpressed in T47D and MDA-MB-453 cells (Figure 1k and Supplementary Figure S2f), suggesting that p16, p27 and p53 play crucial roles in CRB3-mediated regulation of contact inhibition. Cyclin D1 and Cyclin A expression were increased in CRB3 knockdown cells but reduced in CRB3-overexpressing T47D and MDA-MB-453 cells (Figure 1k and Supplementary Figure S2f). Thus, CRB3 is an important factor that regulates contact inhibition.
CRB3 overexpression promotes breast cancer cell apoptosis
We further investigated whether CRB3 overexpression affected cell apoptosis to determine whether contact inhibition was mediated by increased apoptosis or decreased cell proliferation. CRB3 knockdown with siRNA in MCF10A cells had no effect on cell apoptosis (Figures 2a and b). However, CRB3 overexpression in T47D and MDA-MB-453 cells caused a significant increase in apoptosis (Figures 2a and b and Supplementary Figures S4a–b), suggesting that CRB3 overexpression induced cell apoptosis. A three-dimensional (3D) culture model is more suitable than cell monolayers to describe cell apoptosis in MCF10A cells because those can form polarized acinar structures in 3D cultures that are similar to the formation of mammary acini in vivo.29 Therefore, we established a 3D cell culture system to detect apoptosis. Negative control cells formed acini with hollow lumens, whereas CRB3 knockdown (siCRB3-1 and siCRB3-2) resulted in the formation of much larger acini and multiacinar structures (Figures 2c and d). An analysis of cells undergoing apoptosis showed cleaved caspase-3 staining in negative control cells but no staining in CRB3 knockdown cells in 3D cultures (Figure 2e), indicating that CRB3 plays a critical role in regulating the formation of acini. Caspase 3/9, PARP and other apoptosis-related proteins were examined to reveal the detailed mechanism by which CRB3 induced apoptosis. We observed increased levels of the cleaved caspase 3/9 and PARP proteins in CRB3-overexpressing T47D and MDA-MB-453 cells but the levels of these proteins were not increased in the MCF10A cells (Figure 2f and Supplementary Figure S4c). The expression levels of Bcl2 and Survivin were increased and Bad was decreased when CRB3 expression was inhibited in MCF10A cells (Figure 2f). By contrast, Bcl2 and Survivin expression were decreased and Bad expression was increased in CRB3-overexpressing T47D and MDA-MB-453 cells (Figure 2f and Supplementary Figure S4c). Thus, CRB3 may regulate contact inhibition by promoting cell apoptosis.
The loss of CRB3 disrupts the formation of tight junctions and adherens junctions
Cell–cell contacts, including tight and adherens junctions,21, 30 play a crucial role in the maintenance of apical-basal polarity and contact inhibition.5, 31, 32 CRB3 knockdown significantly reduced the adhesion of MCF10A cells in a cell adhesion assay (Figures 3a and b). An immunoblot analysis was performed to determine the expression levels of the tight junction proteins Claudin-1 and ZO-1 and the adherens junction protein E-cadherin. The expression levels of the three proteins were reduced upon CRB3 knockdown, but their expression increased upon CRB3 overexpression (Figure 3c). We investigated the effects of CRB3 knockdown on the localization of ZO-1 and E-cadherin to verify this result. Staining for ZO-1 and E-cadherin in vector control cells revealed a smooth, continuous and well-defined staining pattern at the membranes between cells (Figures 3d and e). By contrast, CRB3 knockdown cells displayed diffuse ZO-1 and E-cadherin staining patterns, suggesting impaired formation of tight and adherens junctions (Figures 3d and e). The 3D culture assay yielded consistent results for E-cadherin localization (Figure 3f). Based on these results, CRB3 regulated contact inhibition by affecting the formation of tight and adherens junctions.
CRB3 is an upstream regulator of the Hippo pathway and affects FRMD6 transcription
The Hippo pathway is implicated in the regulation of contact inhibition.5, 6, 7 CRB3 has previously been shown to be associated with contact inhibition and regulates the Hippo pathway effector YAP.21, 25, 26 Based on these reports, we wondered whether CRB3 was an upstream regulator of the Hippo pathway and whether CRB3 regulated contact inhibition via the Hippo pathway. We first examined the protein expression levels of components of the Hippo pathway in MCF10A and T47D cells grown under sparse and confluent conditions. CRB3 expression was reduced and p-Mst1/2, p-Lats1 and p-YAP expression were increased in cells grown under confluent conditions compared with expression in MCF10A cells grown under sparse conditions (Figure 4a). CRB3 expression was not detected and the YAP and p-YAP expression levels were apparently increased in T47D cells grown under confluent conditions (Figure 4a). The expression levels of Mst1, Mst2, Sav1, Lats1 and Mob1 were not significantly altered in MCF10A and T47D cells (Figure 4a). Lats2 expression was not detected in MCF10A cells since it does not functionally antagonize YAP in these cells.33 As shown in the immunofluorescence analysis of MCF10A cells, YAP was localized to the nucleus in sparse cells and to the cytoplasm in confluent cells (Figure 4b). The YAP protein predominantly remained in the nucleus of T47D cells, even under confluent conditions (Figure 4b). Thus, the Hippo pathway was active in confluent MCF10A cells but was inactive in confluent T47D cells. We then studied the effects of CRB3 on the Hippo pathway. The levels of phosphorylated Mst1/2, Lats1, Mob1 and YAP were decreased, but no changes in the expression of Mst1, Mst2, Lats1 and Mob1 were detected in shCRB3 cells (Figure 4c). By contrast, the levels of phosphorylated Mst1/2, Lats1, Mob1 and YAP were increased in the CRB3-overexpressing cells (Figure 4c). Moreover, p-YAP expression was reduced when Mst2 or Lats1 were knocked down with siRNA in shCRB3 cells (Supplementary Figures S5a and b). The levels of p-Lats1 were decreased when Mst2 was silenced in shCRB3 cells (Supplementary Figure S5a). Thus, CRB3 promoted YAP phosphorylation through the canonical Hippo pathway. The proteins assessed here were proteins downstream of the Hippo pathway, suggesting that CRB3 may regulate the Hippo pathway via upstream proteins.
Kibra and FRMD6, upstream regulators of the Hippo pathway, were previously suggested to regulate the Hippo pathway in MCF10A cells, but NF2 does not act in the same manner.34, 35 Therefore, only FRMD6 and Kibra expression were examined in this study. The levels of the FRMD6 mRNA and protein were downregulated in shCRB3 cells and up-regulated in CRB3-overexpressing cells (Figures 4d–f). The expression of the Kibra protein was positively correlated with CRB3 expression; however, CRB3 had no effect on the expression of the Kibra mRNA (Figures 4d–f). Thus, CRB3 is an upstream regulator of the Hippo pathway and affects FRMD6 transcription.
As shown in the cellular fractionation experiments, nuclear YAP expression was increased in the shCRB3 cells whereas YAP expression was decreased in the CRB3-overexpressing T47D cells (Figure 4g). YAP was localized in the cytoplasm in confluent vector control cells, whereas YAP was localized in the nucleus and cytoplasm in shCRB3 cells, regardless of cell density (Figure 4h). The expression levels of the CTGF and CYR61 mRNAs, two YAP target genes,36, 37 were increased in the shCRB3 cells and inhibited by YAP silencing in the shCRB3 cells (Figure 4i), suggesting that the function of CRB3 may depend on YAP expression. Effective knockdown of YAP expression was confirmed by real-time PCR (Supplementary Figure S6). However, the expression of the CTGF and CYR61 mRNAs was decreased in the CRB3-overexpressing cells (Figure 4j). Thus, CRB3 regulates YAP localization in response to cell density and CRB3 is an upstream regulator of the Hippo pathway.
CRB3 stabilizes Kibra by inhibiting its degradation, and Kibra rescues cell proliferation induced by CRB3 deregulation
A significant change in Kibra mRNA expression was not observed in CRB3-overexpressing cells; thus, we hypothesized that the increased expression of the Kibra protein may be due to the stabilization of the protein. We first measured the half-lives of Kibra and FRMD6 in the context of loss or overexpression of CRB3 to verify this hypothesis. The stability of the FRMD6 protein was not altered by the presence or absence of CRB3 (Figures 5a and b). CRB3 knockdown decreased the half-life of Kibra, whereas CRB3 overexpression significantly prolonged the half-life of Kibra, indicating that CRB3 stabilized the Kibra protein (Figures 5a and b). Like many other proteins, Kibra is degraded through the proteasome pathway. Because the proteasome inhibitor MG132 significantly inhibited the decrease in the levels of the Kibra protein in CRB3 knockdown MCF10A cells and T47D control cells (Figures 5a and b). As shown in immunoprecipitation experiments, CRB3 interacted with Kibra (Figures 5c–f). Interestingly, Kibra was polyubiquitinated in control T47D cells and not in CRB3-overexpressing cells (Figure 5f), suggesting that CRB3 stabilized the Kibra protein by inhibiting its degradation through the ubiquitin-proteasome pathway.
Kibra has previously been shown to regulate YAP phosphorylation.38 We hypothesized that the aforementioned phenotype was caused by Kibra-induced YAP phosphorylation. We measured the levels of p-YAP and its downstream target genes by immunoblotting to confirm this hypothesis. We used a plasmid to overexpress Kibra in the CRB3 knockdown cells because Kibra expression was decreased in MCF10A cells upon CRB3 silencing. Interestingly, Kibra overexpression reversed the expression patterns of p-YAP and the YAP target gene Survivin39 (Figure 5g). Two shRNAs were used to silence Kibra expression because Kibra expression was increased in CRB3-overexpressing T47D cells (Figure 5h). Upregulation of CRB3 increased YAP phosphorylation and reduced Survivin expression, and these alterations were partially reversed upon Kibra silencing (Figure 5h). Based on these results, YAP phosphorylation was mediated by Kibra, and CRB3 exhibited crosstalk with Kibra in the Hippo pathway. Furthermore, Kibra overexpression significantly inhibited BrdU incorporation (Figures 5i and j) and partially rescued the aberrant formation of MCF10A acini (Figures 5k and l) induced by CRB3 knockdown. By contrast, increased BrdU incorporation was observed when Kibra was silenced in CRB3-overexpressing T47D cells (Figures 5m and n). Overall, Kibra rescued cell proliferation and the aberrant formation of acini induced by CRB3 deregulation.
CRB3 inhibits tumour growth in vivo
CRB3 overexpression inhibited breast cancer cell proliferation and induced cell apoptosis in vitro. We wondered whether CRB3 inhibited tumour growth in vivo. The levels of p-Mst1/2, p-Lats1 and p-YAP were increased and the levels of YAP were decreased in CRB3-overexpressing MDA-MB-231 cells (Figure 6a). Meanwhile, BrdU incorporation was decreased (Figures 6b and c). Based on these results, CRB3 reduced the proliferation of MDA-MB-231 cells. In the xenograft studies, the tumour volumes and weights were reduced in the LV-CRB3 group (Figures 6d and e). Necrosis of the LV-CRB3 tumours was increased compared with that of the vector control tumours (Figure 6f). We speculated that necrosis may be due to the increased cell apoptosis because the expression of the apoptosis-related protein PARP was increased in the LV-CRB3 tumours around the necrotic tissue (Figure 6g). Based on these data, CRB3 decreased tumour growth and increased tumour cell apoptosis in vivo.
Discussion
The loss of contact inhibition is a hallmark of cancer cells.4 The Hippo pathway is an important regulator of cell-contact inhibition.5
What’s New in the Hippo keygen?
Screen Shot

System Requirements for Hippo keygen
- First, download the Hippo keygen
-
You can download its setup from given links:



